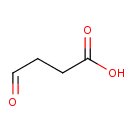
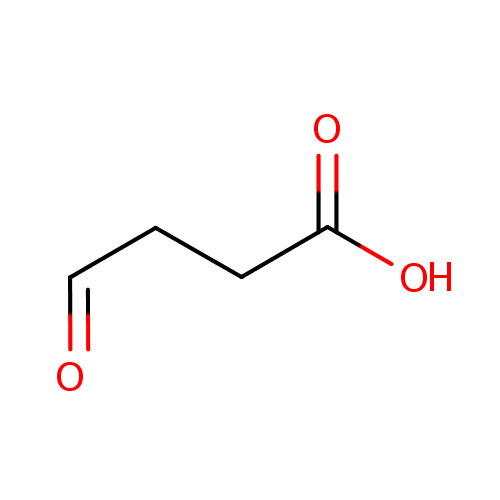
Succinic acid semialdehyde (PAMDB000313)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000313 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | Succinic acid semialdehyde | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | Succinic acid semialdehyde is an intermediate in the catabolism of gamma-aminobutyrate (PMID 16435183). Succinate semialdehyde dehydrogenase is an enzyme that catalyses the reaction of succinate semialdehyde and NAD+ to form succinate and NADH. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C4H6O3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 102.0886 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 102.031694058 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | UIUJIQZEACWQSV-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C4H6O3/c5-3-1-2-4(6)7/h3H,1-2H2,(H,6,7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 692-29-5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | 4-oxobutanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | succinic semialdehyde | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | OC(=O)CCC=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as straight chain fatty acids. These are fatty acids with a straight aliphatic chain. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Lipids and lipid-like molecules | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Fatty Acyls | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Fatty acids and conjugates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Straight chain fatty acids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | Water + NADP + Succinic acid semialdehyde >2 Hydrogen ion + NADPH + Succinic acid gamma-Aminobutyric acid + alpha-Ketoglutarate <> L-Glutamate + Succinic acid semialdehyde Water + NAD + Succinic acid semialdehyde >2 Hydrogen ion + NADH + Succinic acid Hydrogen ion + NADH + Succinic acid semialdehyde <> gamma-Hydroxybutyrate + NAD Oxoglutaric acid + gamma-Aminobutyric acid <> L-Glutamate + Succinic acid semialdehyde Succinic acid semialdehyde + NADP + Water > Succinic acid + NADPH Succinic acid semialdehyde + NAD(P)(+) + Water > Succinic acid + NAD(P)H Succinic acid semialdehyde + NAD + NADP + Water <> Succinic acid + NADH + NADPH +2 Hydrogen ion 4-Hydroxybutanoic acid + NAD <> Succinic acid semialdehyde + NADH + Hydrogen ion gamma-Aminobutyric acid + Oxoglutaric acid > Succinic acid semialdehyde + L-Glutamic acid + L-Glutamate Succinic acid semialdehyde + Water + NADP > NADPH +2 Hydrogen ion + Succinic acid + NADPH | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Bruce, R.; Sims, K.; Pitts, F. N., Jr. Synthesis and purification of succinic semialdehyde. Analytical Biochemistry (1971), 41(1), 271-3. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in 4-aminobutyrate transaminase activity
- Specific function:
- 4-aminobutanoate + 2-oxoglutarate = succinate semialdehyde + L-glutamate
- Gene Name:
- gabT
- Locus Tag:
- PA0266
- Molecular weight:
- 45.2 kDa
Reactions
| 4-aminobutanoate + 2-oxoglutarate = succinate semialdehyde + L-glutamate. |
| (S)-3-amino-2-methylpropanoate + 2-oxoglutarate = 2-methyl-3-oxopropanoate + L-glutamate. |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Succinate semialdehyde + NAD(P)(+) + H(2)O = succinate + NAD(P)H
- Gene Name:
- gabD
- Locus Tag:
- PA0265
- Molecular weight:
- 51.6 kDa
Reactions
| Succinate semialdehyde + NADP(+) + H(2)O = succinate + NADPH. |