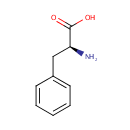
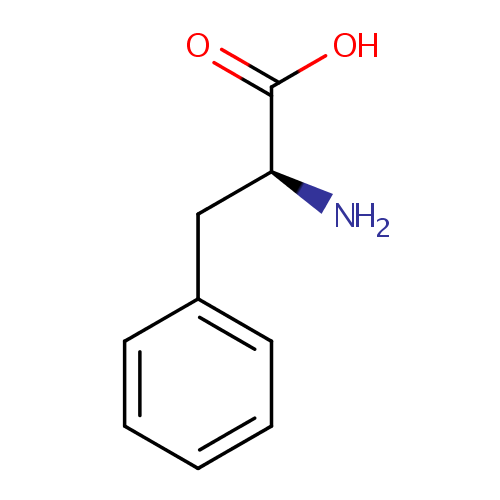
L-Phenylalanine (PAMDB000061)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000061 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | L-Phenylalanine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | Phenylalanine is an essential amino acid and the precursor for the amino acid tyrosine. Phenylalanine is a precursor of the neurotransmitters called catecholamines, which are adrenalin-like substances. Normal metabolism of phenylalanine requires biopterin, iron, niacin, vitamin B6, copper and vitamin C. Phenylalanine and tyrosine, like L-dopa, produce a catecholamine effect. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C9H11NO2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 165.1891 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 165.078978601 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | COLNVLDHVKWLRT-QMMMGPOBSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C9H11NO2/c10-8(9(11)12)6-7-4-2-1-3-5-7/h1-5,8H,6,10H2,(H,11,12)/t8-/m0/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 63-91-2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | (2S)-2-amino-3-phenylpropanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | L-phenylalanine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | N[C@@H](CC1=CC=CC=C1)C(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as phenylpropanoic acids. These are compounds with a structure containing a benzene ring conjugated to a propanoic acid. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Phenylpropanoids and polyketides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Phenylpropanoic acids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Phenylpropanoic acids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic homomonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | 283 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | Adenosine triphosphate + L-Phenylalanine + tRNA(Phe) + tRNA(Phe) <> Adenosine monophosphate + L-Phenylalanyl-tRNA(Phe) + Pyrophosphate + L-Phenylalanyl-tRNA(Phe) alpha-Ketoglutarate + L-Phenylalanine <> L-Glutamate + Phenylpyruvic acid L-Phenylalanine + Oxygen <> 2-Phenylacetamide + Carbon dioxide Adenosine triphosphate + L-Phenylalanine + tRNA(Phe) <> Adenosine monophosphate + Pyrophosphate + L-Phenylalanyl-tRNA(Phe) Phenylpyruvic acid + L-Glutamate <> L-Phenylalanine + Oxoglutaric acid L-Phenylalanine + Adenosine triphosphate + Hydrogen ion + tRNA(Phe) + L-Phenylalanine > Adenosine monophosphate + Pyrophosphate + L-phenylalanyl-tRNA(Phe) Phenylpyruvic acid + L-Glutamic acid + L-Glutamate > Oxoglutaric acid + L-Phenylalanine + L-Phenylalanine L-Phenylalanine + Oxygen + L-Phenylalanine <> Oxoglutaric acid + Phenylpyruvic acid L-Phenylalanine + Oxygen + L-Phenylalanine <> Carbon dioxide + Sinapyl alcohol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Zhou, Hua; Zhong, Yao; Sun, Guanghai; Wei, Ping. Preparation of L-phenylalanine by an aqueous two-phase system. Huaxue Fanying Gongcheng Yu Gongyi (2006), 22(2), 146-150. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in transferase activity
- Specific function:
- An aromatic amino acid + 2-oxoglutarate = an aromatic oxo acid + L-glutamate
- Gene Name:
- tyrB
- Locus Tag:
- PA3139
- Molecular weight:
- 43.3 kDa
Reactions
| An aromatic amino acid + 2-oxoglutarate = an aromatic oxo acid + L-glutamate. |
- General function:
- Involved in RNA binding
- Specific function:
- ATP + L-phenylalanine + tRNA(Phe) = AMP + diphosphate + L-phenylalanyl-tRNA(Phe)
- Gene Name:
- pheT
- Locus Tag:
- PA2739
- Molecular weight:
- 86.8 kDa
Reactions
| ATP + L-phenylalanine + tRNA(Phe) = AMP + diphosphate + L-phenylalanyl-tRNA(Phe). |
- General function:
- Involved in nucleotide binding
- Specific function:
- ATP + L-phenylalanine + tRNA(Phe) = AMP + diphosphate + L-phenylalanyl-tRNA(Phe)
- Gene Name:
- pheS
- Locus Tag:
- PA2740
- Molecular weight:
- 38.1 kDa
Reactions
| ATP + L-phenylalanine + tRNA(Phe) = AMP + diphosphate + L-phenylalanyl-tRNA(Phe). |
- General function:
- Involved in chorismate mutase activity
- Specific function:
- Chorismate = prephenate
- Gene Name:
- pheA
- Locus Tag:
- PA3166
- Molecular weight:
- 40.6 kDa
Reactions
| Chorismate = prephenate. |
| Prephenate = phenylpyruvate + H(2)O + CO(2). |
- General function:
- Involved in catalytic activity
- Specific function:
- Acts on leucine, isoleucine and valine
- Gene Name:
- ilvE
- Locus Tag:
- PA5013
- Molecular weight:
- 34.1 kDa
Reactions
| L-leucine + 2-oxoglutarate = 4-methyl-2-oxopentanoate + L-glutamate. |
| L-isoleucine + 2-oxoglutarate = (S)-3-methyl-2-oxopentanoate + L-glutamate. |
| L-valine + 2-oxoglutarate = 3-methyl-2-oxobutanoate + L-glutamate. |
Transporters
- General function:
- Involved in nucleotide binding
- Specific function:
- Probably part of a binding-protein-dependent transport system yecCS for an amino acid. Probably responsible for energy coupling to the transport system
- Gene Name:
- yecC
- Locus Tag:
- PA5152
- Molecular weight:
- 28.4 kDa
- General function:
- Involved in transport
- Specific function:
- Permease that is involved in the transport across the cytoplasmic membrane of the aromatic amino acids (phenylalanine, tyrosine, and tryptophan)
- Gene Name:
- aroP
- Locus Tag:
- PA3000
- Molecular weight:
- 51 kDa