|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB000654 |
|---|
|
Identification |
|---|
| Name: |
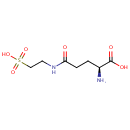
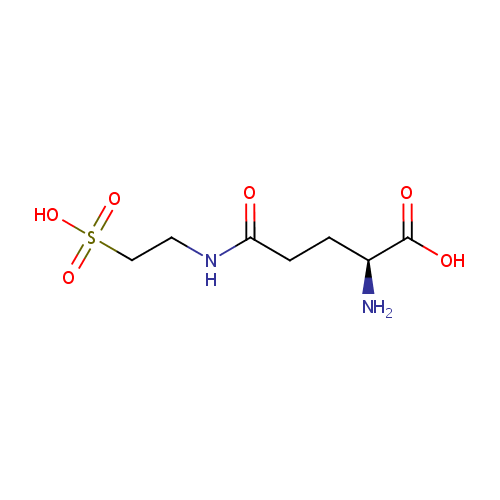
5-L-Glutamyl-taurine |
|---|
| Description: | 5-L-Glutamyl-taurine is an intermediate in taurine and hypotaurine metabolism. 5-L-Glutamyl-taurine is produced from Taurine via the enzyme gamma-glutamyltranspeptidase (EC 2.3.2.2). |
|---|
|
Structure |
|
|---|
| Synonyms: | |
|---|
|
Chemical Formula: |
C7H14N2O6S |
|---|
| Average Molecular Weight: |
254.261 |
|---|
| Monoisotopic Molecular
Weight: |
254.05725688 |
|---|
| InChI Key: |
WGXUDTHMEITUBO-YFKPBYRVSA-N |
|---|
| InChI: | InChI=1S/C7H14N2O6S/c8-5(7(11)12)1-2-6(10)9-3-4-16(13,14)15/h5H,1-4,8H2,(H,9,10)(H,11,12)(H,13,14,15)/t5-/m0/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | (2S)-2-amino-4-[(2-sulfoethyl)carbamoyl]butanoic acid |
|---|
|
Traditional IUPAC Name: |
(2S)-2-amino-4-[(2-sulfoethyl)carbamoyl]butanoic acid |
|---|
| SMILES: | N[C@@H](CCC(=O)NCCS(O)(=O)=O)C(O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as l-alpha-amino acids. These are alpha amino acids which have the L-configuration of the alpha-carbon atom. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
|
Class |
Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
|
Direct Parent |
L-alpha-amino acids |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- L-alpha-amino acid
- Amino fatty acid
- Fatty acyl
- Fatty acid
- N-acyl-amine
- Fatty amide
- Alkanesulfonic acid
- Sulfonyl
- Sulfonic acid derivative
- Sulfonic acid
- Secondary carboxylic acid amide
- Carboxamide group
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid amide
- Hydrocarbon derivative
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Carbonyl group
- Amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
- Taurine and hypotaurine metabolism pae00430
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | HMDB04195 | | Pubchem Compound ID | 68759 | | Kegg ID | C05844 | | ChemSpider ID | 62003 | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|