|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB110828 |
|---|
|
Identification |
|---|
| Name: |
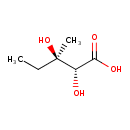
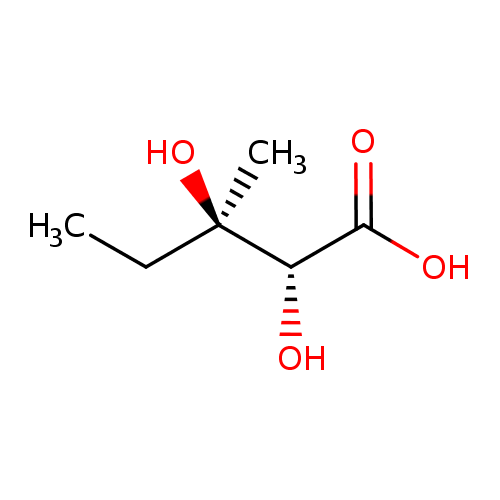
(R)-2,3-dihydroxy-3-methylpentanoate |
|---|
| Description: | (R) 2,3-Dihydroxy-methylvalerate is an intermediate in valine, leucine and isoleucine biosynthesis. The pathway of valine biosynthesis is a four-step pathway that shares all of its steps with the parallel pathway of isoleucine biosynthesis. These entwined pathways are part of the superpathway of leucine, valine, and isoleucine biosynthesis, that generates not only isoleucine and valine, but also leucine. (R) 2,3-Dihydroxy-methylvalerate is generated from 3-Hydroxy-3-methyl-2-oxopentanoic acid via the enzyme ketol-acid reductoisomerase (EC 1.1.1.86) then it is converted to (S)-3-methyl-2-oxopentanoic via the dihydroxy-acid dehydratase (EC:4.2.1.9). |
|---|
|
Structure |
|
|---|
| Synonyms: | -
(R)-2,3-dihydroxy-3-methylvalerate
|
|---|
|
Chemical Formula: |
C6H11O4
|
|---|
| Average Molecular Weight: |
147.15 |
|---|
| Monoisotopic Molecular
Weight: |
148.0735588736 |
|---|
| InChI Key: |
PDGXJDXVGMHUIR-UJURSFKZSA-M |
|---|
| InChI: |
InChI=1S/C6H12O4/c1-3-6(2,10)4(7)5(8)9/h4,7,10H,3H2,1-2H3,(H,8,9)/p-1/t4-,6+/m0/s1 |
|---|
| CAS
number: |
562-43-6 |
|---|
| IUPAC Name: | (2R,3R)-2,3-dihydroxy-3-methylpentanoic acid |
|---|
|
Traditional IUPAC Name: |
(2R,3R)-2,3-dihydroxy-3-methylpentanoic acid |
|---|
| SMILES: | CCC(O)(C)C(C([O-])=O)O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as hydroxy fatty acids. These are fatty acids in which the chain bears a hydroxyl group. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Lipids and lipid-like molecules |
|---|
| Sub Class | Fatty Acyls |
|---|
|
Direct Parent |
Hydroxy fatty acids |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Branched fatty acid
- Methyl-branched fatty acid
- Short-chain hydroxy acid
- Hydroxy fatty acid
- Alpha-hydroxy acid
- Hydroxy acid
- Monosaccharide
- Tertiary alcohol
- 1,2-diol
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Organooxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|