|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB110757 |
|---|
|
Identification |
|---|
| Name: |
butanoate |
|---|
| Description: | A short-chain fatty acid anion that is the conjugate base of butyric acid, obtained by deprotonation of the carboxy group. |
|---|
|
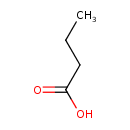
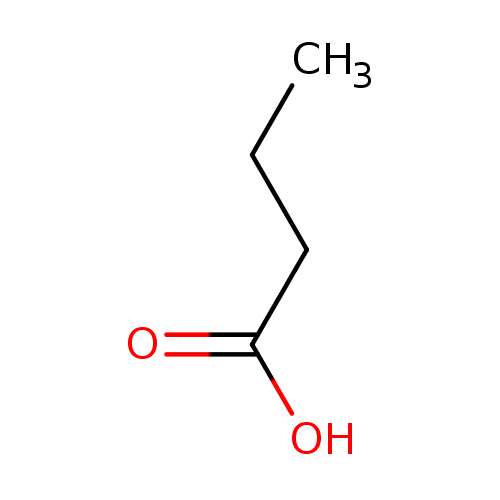
Structure |
|
|---|
| Synonyms: | -
butyrate
-
butyric acid
-
butanoic acid
-
ethylacetic acid
-
n-butanoate
-
1-propanecarboxylic acid
-
propylformic acid
|
|---|
|
Chemical Formula: |
C4H7O2
|
|---|
| Average Molecular Weight: |
87.098 |
|---|
| Monoisotopic Molecular
Weight: |
88.052429501 |
|---|
| InChI Key: |
FERIUCNNQQJTOY-UHFFFAOYSA-M |
|---|
| InChI: |
InChI=1S/C4H8O2/c1-2-3-4(5)6/h2-3H2,1H3,(H,5,6)/p-1 |
|---|
| CAS
number: |
107-92-6 |
|---|
| IUPAC Name: | butanoate |
|---|
|
Traditional IUPAC Name: |
butyric acid |
|---|
| SMILES: | CCCC(=O)[O-] |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as straight chain fatty acids. These are fatty acids with a straight aliphatic chain. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Lipids and lipid-like molecules |
|---|
| Sub Class | Fatty Acyls |
|---|
|
Direct Parent |
Straight chain fatty acids |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Straight chain fatty acid
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Liquid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
-5.7 °C |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | -5.7 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 60.0 mg/mL | Not Available | | LogP | 0.79 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
| Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS | Not Available |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-000i-9000000000-7f461db56bfd8568ec71 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-000i-9000000000-66f857fa612f773837bc | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-000i-9000000000-e6689b2e6bf21570b934 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI RMU-7M) , Positive | splash10-03dl-9000000000-b2ffa7d67b2466dea94f | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80B) , Positive | splash10-03dl-9000000000-7467bf19c64fd3f51105 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-000i-9000000000-9ae015043b014b3c93d9 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-000i-9000000000-e30b3c6bd6218b9b49e1 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-000i-9000000000-efbb0e35a19a1713240b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-9000000000-acb5cf0017a9ee680dd8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dl-9000000000-812e24462a71dccb0fec | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-f9d30338ca1ee9409964 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-9000000000-9d749b6b6cf2f93a8f85 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00ku-9000000000-ee742730266fb4997777 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-88fc445cddcb726e93d8 | View in MoNA |
|---|
| MS | Mass Spectrum (Electron Ionization) | splash10-03di-9000000000-5338ff8a9c4e59150aba | View in MoNA |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available |
|---|
| 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available |
|---|
|
|---|
|
References |
|---|
| References: |
- Lupton JR (1995)Butyrate and colonic cytokinetics: differences between in vitro and in vivo studies. European journal of cancer prevention : the official journal of the European Cancer Prevention Organisation (ECP) 4, Pubmed: 7496326
- Salek RM, Maguire ML, Bentley E, Rubtsov DV, Hough T, Cheeseman M, Nunez D, Sweatman BC, Haselden JN, Cox RD, Connor SC, Griffin JL (2007)A metabolomic comparison of urinary changes in type 2 diabetes in mouse, rat, and human. Physiological genomics 29, Pubmed: 17190852
|
|---|
| Synthesis Reference: |
Shan, Zhiping. Preparation of butyric acid by hydrogenation of maleic anhydride. U.S. Pat. Appl. Publ. (2008), 6pp. |
|---|
| Material Safety Data Sheet (MSDS) |
Download (PDF) |
|---|
|
Links |
|---|
| External Links: |
|
|---|