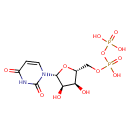
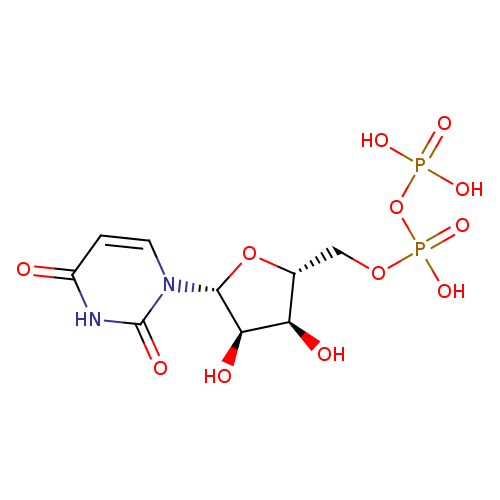
UDP (PAMDB110529)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB110529 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | UDP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | An organophosphate oxoanion arising from deprotonation of the diphosphate OH groups of uridine 5'-diphosphate; major species at pH 7.3. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C9H11N2O12P2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 401.14 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 404.002196945 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | XCCTYIAWTASOJW-XVFCMESISA-K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C9H14N2O12P2/c12-5-1-2-11(9(15)10-5)8-7(14)6(13)4(22-8)3-21-25(19,20)23-24(16,17)18/h1-2,4,6-8,13-14H,3H2,(H,19,20)(H,10,12,15)(H2,16,17,18)/p-3/t4-,6-,7-,8-/m1/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 58-98-0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | 5'-O-[(phosphonatooxy)phosphinato]uridine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | UDP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | C(OP(=O)([O-])OP(=O)([O-])[O-])C1(OC(C(O)C(O)1)N2(C=CC(=O)NC(=O)2)) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of chemical entities known as pyrimidine ribonucleoside diphosphates. These are pyrimidine ribonucleotides with diphosphate group linked to the ribose moiety. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Chemical entities | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Nucleosides, nucleotides, and analogues | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Pyrimidine nucleotides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Pyrimidine ribonucleoside diphosphates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic heteromonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | UDP-α-D-glucose + β-D-fructofuranose 6-phosphate → sucrose 6F-phosphate + UDP + Hydrogen ion Uridine 5''-diphospho-{beta}-4-deoxy-4-formamido-L-arabinose + di-trans,octa-cis-undecaprenyl phosphate → 4-deoxy-4-formamido-α-L-arabinopyranosyl ditrans,octacis-undecaprenyl phosphate + UDP UDP-N-acetyl-α-D-glucosamine + Water → N-acetyl-α-D-mannosamine + UDP + Hydrogen ion decaprenyl-diphospho-N-acetylmuramoyl-pentapeptide + UDP-N-acetyl-α-D-glucosamine → Hydrogen ion + decaprenyl-pyrophosphoryl-(N-acetylglucosamine)-N-acetylmuramyl-(pentapeptide) + UDP undecaprenyl-diphospho-N-acetylmuramoyl-L-alanyl-γ-D-glutamyl-L-lysyl- D-alanine + UDP-N-acetyl-α-D-glucosamine → Hydrogen ion + undecaprenyl-diphospho-(N-acetylglucosamine)-N-acetylmuramoyl-L-alanyl-γ-D-glutamyl-L-lysyl-D-alanine + UDP undecaprenyl-diphospho-N-acetylmuramoyl-L-alanyl-γ-D-glutamyl-L-lysyl- D-alanyl-D-alanine + UDP-N-acetyl-α-D-glucosamine → Hydrogen ion + undecaprenyldiphospho-N-acetyl-(N-acetylglucosaminyl)muramoyl-L-alanyl-γ-D-glutamyl-L-lysyl-D-alanyl-D-alanine + UDP UDP-α-D-glucose + D-Fructose → Sucrose + UDP + Hydrogen ion UDP-α-D-glucose + galactosyl-(glucosyl)2-(heptosyl)3-Kdo2-lipid A-bisphosphate → Hydrogen ion + galactosyl-(glucosyl)3-(heptosyl)3-Kdo2-lipid A-bisphosphate + UDP UDP-α-D-glucose + galactosyl-glucosyl-(heptosyl)3-Kdo2-lipid A-bisphosphate → Hydrogen ion + galactosyl-(glucosyl)2-(heptosyl)3-Kdo2-lipid A-bisphosphate + UDP UDP-α-D-glucose + Glucopyranose → Trehalose + UDP + Hydrogen ion UDP-α-D-galactose + glucosyl-(heptosyl)3-Kdo2-lipid A-bisphosphate → Hydrogen ion + galactosyl-glucosyl-(heptosyl)3-Kdo2-lipid A-bisphosphate + UDP UDP-α-D-glucose + (heptosyl)2-Kdo2-lipid A → Hydrogen ion + glucosyl-(heptosyl)2-Kdo2-lipid A + UDP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Zeng, Bin; Rao, Linfan; Li, Gaowo. Method for manufacturing uridine diphosphate. Faming Zhuanli Shenqing Gongkai Shuomingshu (2007), 14pp. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||