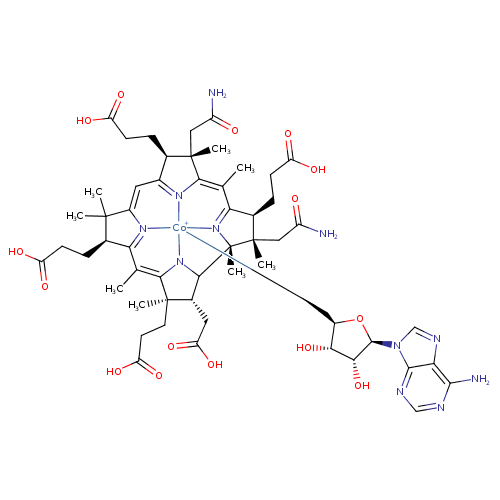
adenosyl-cobyrinate a,c-diamide (PAMDB110418)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB110418 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | adenosyl-cobyrinate a,c-diamide | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | Conjugate base of adenosylcob(III)yrinic acid a,c-diamide. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C55H68N11O15Co | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 1182.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 1186.461960932 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | OCNLJCZKGHKJGF-NQYRMHKHSA-H | |||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C45H62N6O12.C10H12N5O3.Co/c1-21-36-24(10-13-32(56)57)41(3,4)28(49-36)18-27-23(9-12-31(54)55)43(6,19-29(46)52)39(48-27)22(2)37-25(11-14-33(58)59)44(7,20-30(47)53)45(8,51-37)40-26(17-35(62)63)42(5,38(21)50-40)16-15-34(60)61;1-4-6(16)7(17)10(18-4)15-3-14-5-8(11)12-2-13-9(5)15;/h18,23-26,40H,9-17,19-20H2,1-8H3,(H10,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63);2-4,6-7,10,16-17H,1H2,(H2,11,12,13);/q;;+2/p-6/t23-,24-,25-,26+,40-,42-,43+,44+,45+;4-,6-,7-,10-;/m11./s1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | CC5(=C%10(C(C)(CCC([O-])=O)C(CC(=O)[O-])C9(C%11(C)(C(C)(CC(N)=O)C(CCC([O-])=O)C1(=[N+]([Co--]3([N+]2(C(=C(C)1)C(C)(CC(N)=O)C(CCC([O-])=O)C=2C=C4(C(C)(C)C(CCC([O-])=O)C(=[N+]34)5)))(CC6(C(C(O)C(O6)N7(C=NC8(=C7N=CN=C8N)))O))N9%10)%11))))) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as metallotetrapyrroles. These are polycyclic compounds containing a tetrapyrrole skeleton combined with a metal atom. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organoheterocyclic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Tetrapyrroles and derivatives | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Metallotetrapyrroles | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Metallotetrapyrroles | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic heteropolycyclic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -4 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| |||||||||||||||||||||||||||||||||||||||||||||||||||