|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB110071 |
|---|
|
Identification |
|---|
| Name: |
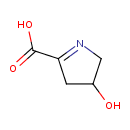
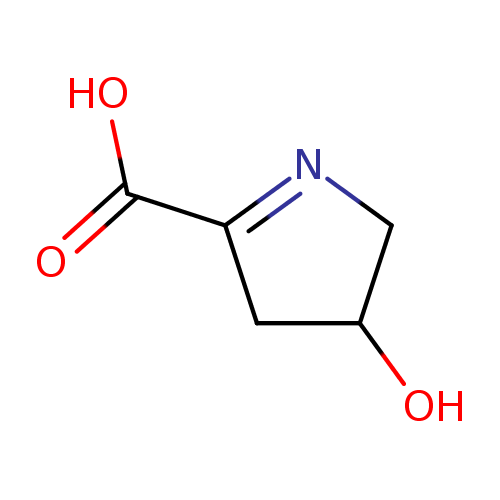
1-pyrroline-4-hydroxy-2-carboxylate |
|---|
| Description: | The conjugate base of 4-hydroxy-1-pyrroline-2-carboxylic acid; major species at pH 7.3. |
|---|
|
Structure |
|
|---|
| Synonyms: | |
|---|
|
Chemical Formula: |
C5H6NO3
|
|---|
| Average Molecular Weight: |
129.0425930962 |
|---|
| Monoisotopic Molecular
Weight: |
129.0425930962 |
|---|
| InChI Key: |
AOMLMYXPXUTBQH-UHFFFAOYSA-M |
|---|
| InChI: |
InChI=1S/C5H7NO3/c7-3-1-4(5(8)9)6-2-3/h3,7H,1-2H2,(H,8,9)/p-1 |
|---|
| CAS
number: |
9054-77-7 |
|---|
| IUPAC Name: | 3-hydroxy-3,4-dihydro-2H-pyrrole-5-carboxylate |
|---|
|
Traditional IUPAC Name: |
4-hydroxy-4,5-dihydro-3H-pyrrole-2-carboxylic acid |
|---|
| SMILES: | C1(N=C(C([O-])=O)CC(O)1) |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as pyrrolines. These are compounds containing a pyrroline ring, which is a five-member unsaturated aliphatic ring with one nitrogen atom and four carbon atoms. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Organoheterocyclic compounds |
|---|
| Sub Class | Pyrrolines |
|---|
|
Direct Parent |
Pyrrolines |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Pyrroline
- Ketimine
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Azacycle
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Alcohol
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Imine
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
- Arginine and Proline Metabolism pae00330
|
|---|
|
Spectra |
|---|
| Spectra: |
Not Available |
|---|
|
References |
|---|
| References: |
- Heacock AM, Adams E: Formation and excretion of pyrrole-2-carboxylate in man. J Clin Invest. 1974 Oct;54(4):810-8. [4430715 ]
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|