|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB110029 |
|---|
|
Identification |
|---|
| Name: |
S-ureidoglycolate |
|---|
| Description: | Conjugate base of (−)-ureidoglycolic acid. |
|---|
|
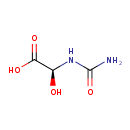
Structure |
|
|---|
| Synonyms: | -
(-)-ureidoglycolate
-
ureidoglycolate
-
S-(-)-ureidoglycolate
|
|---|
|
Chemical Formula: |
C3H5N2O4
|
|---|
| Average Molecular Weight: |
133.08 |
|---|
| Monoisotopic Molecular
Weight: |
134.0327566914 |
|---|
| InChI Key: |
NWZYYCVIOKVTII-SFOWXEAESA-M |
|---|
| InChI: |
InChI=1S/C3H6N2O4/c4-3(9)5-1(6)2(7)8/h1,6H,(H,7,8)(H3,4,5,9)/p-1/t1-/m0/s1 |
|---|
| CAS
number: |
7424-03-5 |
|---|
| IUPAC Name: | (2S)-(carbamoylamino)(hydroxy)acetate |
|---|
|
Traditional IUPAC Name: |
ureidoglycolate |
|---|
| SMILES: | C(O)(C([O-])=O)NC(N)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as n-carbamoyl-alpha amino acids. These are compounds containing an alpha amino acid which bears an carbamoyl group at its terminal nitrogen atom. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Organic acids and derivatives |
|---|
| Sub Class | Carboxylic acids and derivatives |
|---|
|
Direct Parent |
N-carbamoyl-alpha amino acids |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- N-carbamoyl-alpha-amino acid
- Ureide
- Alpha-hydroxy acid
- Hydroxy acid
- Urea
- Alkanolamine
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Carbonyl group
- Organic oxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
- allantoin degradation IV (anaerobic)PWY0-41
- allantoin degradation to ureidoglycolate I (urea producing)PWY-5697
- superpathway of allantoin degradation in plantsURDEGR-PWY
- allantoin degradation to glyoxylate IPWY-5694
- allantoin degradation to ureidoglycolate II (ammonia producing)PWY-5698
- allantoin degradation to glyoxylate IIIPWY-5705
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|