|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB004066 |
|---|
|
Identification |
|---|
| Name: |
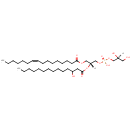
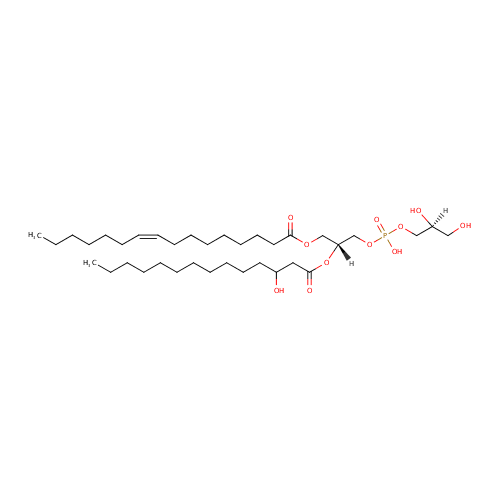
PG(14:0(3-OH)/16:1(9Z)) |
|---|
| Description: | PG(14:0(3-OH)/16:1(9Z)) is a phosphatidylglycerol. Phosphatidylglycerols consist of a glycerol 3-phosphate backbone esterified to either saturated or unsaturated fatty acids on carbons 1 and 2. As is the case with diacylglycerols, phosphatidylglycerols can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 positions. PG(14:0(3-OH)/16:1(9Z)), in particular, consists of one 3-hydroxytetradecanoyl chain to the C-1 atom, and one 9Z-hexadecenoyl to the C-2 atom. In Pseudomonas aeruginosa glycerophospholipid metabolism, phosphatidylglycerol is formed from phosphatidic acid (1,2-diacyl-sn-glycerol 3-phosphate) by a sequence of enzymatic reactions that proceeds via two intermediates, cytidine diphosphate diacylglycerol (CDP-diacylglycerol) and phosphatidylglycerophosphate (PGP, a phosphorylated phosphatidylglycerol). Phosphatidylglycerols, along with CDP-diacylglycerol, also serve as precursor molecules for the synthesis of cardiolipin, a phospholipid found in membranes. |
|---|
|
Structure |
|
|---|
| Synonyms: | - 1-3-hydroxytetradecanoyl-2-(9Z-hexadecenoyl)-sn-glycero-3-phospho-(1'-glycerol)
- 1-3-hydroxytetradecanoyl-2-(9Z-hexadecenoyl)-sn-glycero-3-phosphoglycerol
- 1-3-hydroxytetradecanoyl-2-palmitoleoyl-sn-glycero-3-phosphoglycerol
- GPG(14:0/16:1)
- GPG(30:1)
- PG(14:0/16:1)
- PG(30:1)
- Phosphatidylglycerol(14:0/16:1)
- Phosphatidylglycerol(30:1)
|
|---|
|
Chemical Formula: |
C36H69O11P |
|---|
| Average Molecular Weight: |
708.911 |
|---|
| Monoisotopic Molecular
Weight: |
708.45775004 |
|---|
| InChI Key: |
HKPIQAAQQHSIBQ-GQOIWLODSA-N |
|---|
| InChI: | InChI=1S/C36H69O11P/c1-3-5-7-9-11-13-14-15-16-18-20-22-24-26-35(40)44-30-34(31-46-48(42,43)45-29-33(39)28-37)47-36(41)27-32(38)25-23-21-19-17-12-10-8-6-4-2/h13-14,32-34,37-39H,3-12,15-31H2,1-2H3,(H,42,43)/b14-13-/t32?,33-,34+/m0/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | [(2S)-2,3-dihydroxypropoxy][(2R)-3-[(9Z)-hexadec-9-enoyloxy]-2-[(3-hydroxytetradecanoyl)oxy]propoxy]phosphinic acid |
|---|
|
Traditional IUPAC Name: |
(2S)-2,3-dihydroxypropoxy((2R)-3-[(9Z)-hexadec-9-enoyloxy]-2-[(3-hydroxytetradecanoyl)oxy]propoxy)phosphinic acid |
|---|
| SMILES: | [H][C@](O)(CO)COP(O)(=O)OC[C@@]([H])(COC(=O)CCCCCCC\C=C/CCCCCC)OC(=O)CC(O)CCCCCCCCCCC |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as phosphatidylglycerols. These are glycerophosphoglycerols in which two fatty acids are bonded to the 1-glycerol moiety through ester linkages. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Glycerophospholipids |
|---|
| Sub Class | Glycerophosphoglycerols |
|---|
|
Direct Parent |
Phosphatidylglycerols |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- 1,2-diacylglycerophosphoglycerol
- Beta-hydroxy acid
- Dialkyl phosphate
- Fatty acid ester
- Fatty acyl
- Alkyl phosphate
- Dicarboxylic acid or derivatives
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Hydroxy acid
- 1,2-diol
- Carboxylic acid ester
- Secondary alcohol
- Carboxylic acid derivative
- Primary alcohol
- Hydrocarbon derivative
- Organic oxygen compound
- Carbonyl group
- Organic oxide
- Alcohol
- Organooxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Membrane |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Yuan Y, Leeds JA, Meredith TC (2012). "Pseudomonas aeruginosa Directly Shunts β-Oxidation Degradation Intermediates into De Novo Fatty Acid Biosynthesis." J Bacteriol. 194(19):5185-96 Pubmed: 22753057
- Yurtsever D. (2007). Fatty acid methyl ester profiling of Enterococcus and Esherichia coli for microbial source tracking. M.sc. Thesis. Villanova University: U.S.A
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | Not Available | | Kegg ID | Not Available | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|