|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB003728 |
|---|
|
Identification |
|---|
| Name: |
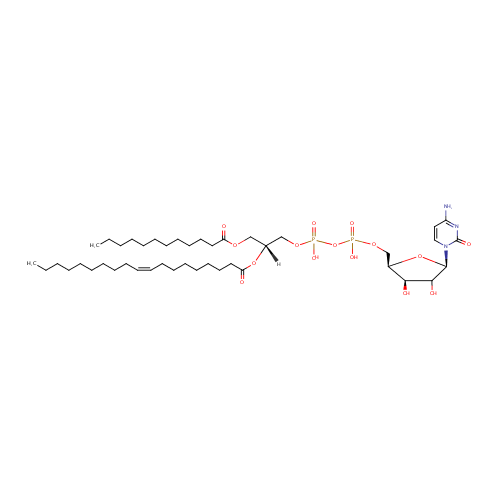
CDP-DG(12:0/19:1(9Z)) |
|---|
| Description: | CDP-DG(12:0/19:1(9Z)) belongs to the family of CDP-diacylglycerols. It is a glycerophospholipid containing a diacylglycerol, with a cytidine diphosphate attached to the oxygen O1 or O2 of the glycerol part. As is the case with diacylglycerols, phosphatidylserines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. CDP-DG(12:0/19:1(9Z)), in particular, consists of one dodecanoyl chain to C-1 atom, and one 9Z-nonadecenoyl to the C-2 atom. In Pseudomonas aeruginosa glycerophospholipid metabolism, The biosynthesis of CDP-diacylglycerol (CDP-DG) involves condensation of phosphatidic acid (PA) and cytidine triphosphate, with elimination of pyrophosphate, catalysed by the enzyme CDP-diacylglycerol synthase. The resulting CDP-diacylglycerol can be utilized immediately for the synthesis of phosphatidylglycerol (PG), and thence cardiolipin (CL), and of phosphatidylinositol (PI). CDP-DG(12:0/19:1(9Z)) is also a substrate of CDP-diacylglycerol pyrophosphatase. It is involved in CDP-diacylglycerol degradation pathway. |
|---|
|
Structure |
|
|---|
| Synonyms: | - 1-dodecanoyl-2-(9Z-nonadecenoyl)-sn-glycero-3-CDP
- 1-dodecanoyl-2-(9Z-nonadecenoyl)-sn-glycero-3-cytidine-5'-diphosphate
- 1-Dodecanoyl-2-(9Z-nonadecenoyl)-sn-glycero-3-cytidine-5'-diphosphoric acid
- 1-dodecanoyl-2-9Z-nonadecenoyl-sn-glycero-3-cytidine-5'-diphosphate
- 1-Dodecanoyl-2-9Z-nonadecenoyl-sn-glycero-3-cytidine-5'-diphosphoric acid
- CDP-DG(12:0/19:1)
- CDP-DG(31:1)
- CDP-Diacylglycerol(12:0/19:1)
- CDP-Diacylglycerol(31:1)
|
|---|
|
Chemical Formula: |
C43H77N3O15P2 |
|---|
| Average Molecular Weight: |
938.043 |
|---|
| Monoisotopic Molecular
Weight: |
937.482992787 |
|---|
| InChI Key: |
NBJRXNPRBWIPKM-DXDDRBQSSA-N |
|---|
| InChI: | InChI=1S/C43H77N3O15P2/c1-3-5-7-9-11-13-14-15-16-17-18-19-21-23-25-27-29-39(48)59-35(32-56-38(47)28-26-24-22-20-12-10-8-6-4-2)33-57-62(52,53)61-63(54,55)58-34-36-40(49)41(50)42(60-36)46-31-30-37(44)45-43(46)51/h16-17,30-31,35-36,40-42,49-50H,3-15,18-29,32-34H2,1-2H3,(H,52,53)(H,54,55)(H2,44,45,51)/b17-16-/t35-,36-,40+,41?,42-/m1/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | {[(2R,3R,5R)-5-(4-amino-2-oxo-1,2-dihydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}({[(2R)-3-(dodecanoyloxy)-2-[(9Z)-nonadec-9-enoyloxy]propoxy](hydroxy)phosphoryl}oxy)phosphinic acid |
|---|
|
Traditional IUPAC Name: |
[(2R,3R,5R)-5-(4-amino-2-oxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy([(2R)-3-(dodecanoyloxy)-2-[(9Z)-nonadec-9-enoyloxy]propoxy(hydroxy)phosphoryl]oxy)phosphinic acid |
|---|
| SMILES: | [H][C@@](COC(=O)CCCCCCCCCCC)(COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H](C(O)[C@H]1O)N1C=CC(N)=NC1=O)OC(=O)CCCCCCC\C=C/CCCCCCCCC |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as cdp-diacylglycerols. These are glycerolipids containing a diacylglycerol, with a cytidine diphosphate attached to the oxygen O1 or O2 of the glycerol part. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Glycerophospholipids |
|---|
| Sub Class | CDP-glycerols |
|---|
|
Direct Parent |
CDP-diacylglycerols |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Cdp-diacylglycerol
- Diacyl-glycerol-3-pyrophosphate
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- Organic pyrophosphate
- Monosaccharide phosphate
- Fatty acid ester
- Hydroxypyrimidine
- Monoalkyl phosphate
- Alkyl phosphate
- Organic phosphoric acid derivative
- Monosaccharide
- Pyrimidine
- Phosphoric acid ester
- Fatty acyl
- Hydropyrimidine
- Dicarboxylic acid or derivatives
- Heteroaromatic compound
- Oxolane
- Secondary alcohol
- 1,2-diol
- Carboxylic acid ester
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aromatic heteromonocyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -2 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Membrane |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- Yurtsever D. (2007). Fatty acid methyl ester profiling of Enterococcus and Esherichia coli for microbial source tracking. M.sc. Thesis. Villanova University: U.S.A
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | Not Available | | Kegg ID | Not Available | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|