|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB003420 |
|---|
|
Identification |
|---|
| Name: |
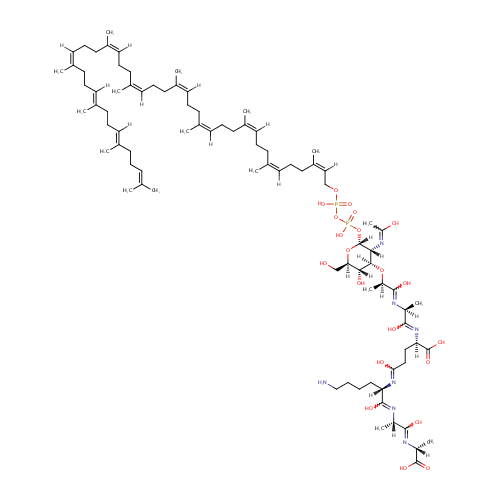
Mur2Ac(oyl-L-Ala-gamma-D-Glu-L-Lys-D-Ala-D-Ala)-diphosphoundecaprenol |
|---|
| Description: | Mur2Ac(oyl-L-Ala-gamma-D-Glu-L-Lys-D-Ala-D-Ala)-diphosphoundecaprenol is an intermediate in peptidoglycan synthesis. It is a substrate for the enzyme undecaprenyldiphospho-muramoylpentapeptide beta-N-acetylglucosaminyltransferase which catalyzes the reaction: UDP-N-acetylglucosamine + Mur2Ac(oyl-L-Ala-gamma-D-Glu-L-Lys-D-Ala-D-Ala)-diphosphoundecaprenol = UDP + N-acetylglucosamine-(1-4)-Mur2Ac(oyl-L-Ala-gamma-D-Glu-L-Lys-D-Ala-D-Ala)-diphosphoundecaprenol. |
|---|
|
Structure |
|
|---|
| Synonyms: | - Mur2ac(oyl-L-ala-g-D-glu-L-lys-D-ala-D-ala)-diphosphoundecaprenol
- Mur2ac(oyl-L-ala-γ-D-glu-L-lys-D-ala-D-ala)-diphosphoundecaprenol
|
|---|
|
Chemical Formula: |
C86H143N7O21P2 |
|---|
| Average Molecular Weight: |
1673.0374 |
|---|
| Monoisotopic Molecular
Weight: |
1671.981227697 |
|---|
| InChI Key: |
SULOOAFLXMQJSF-NDEPDNEDSA-N |
|---|
| InChI: | InChI=1S/C86H143N7O21P2/c1-57(2)30-20-31-58(3)32-21-33-59(4)34-22-35-60(5)36-23-37-61(6)38-24-39-62(7)40-25-41-63(8)42-26-43-64(9)44-27-45-65(10)46-28-47-66(11)48-29-49-67(12)53-55-110-115(106,107)114-116(108,109)113-86-77(91-72(17)95)79(78(97)75(56-94)112-86)111-71(16)82(100)88-69(14)81(99)93-74(85(104)105)51-52-76(96)92-73(50-18-19-54-87)83(101)89-68(13)80(98)90-70(15)84(102)103/h30,32,34,36,38,40,42,44,46,48,53,68-71,73-75,77-79,86,94,97H,18-29,31,33,35,37,39,41,43,45,47,49-52,54-56,87H2,1-17H3,(H,88,100)(H,89,101)(H,90,98)(H,91,95)(H,92,96)(H,93,99)(H,102,103)(H,104,105)(H,106,107)(H,108,109)/b58-32+,59-34+,60-36-,61-38-,62-40-,63-42-,64-44-,65-46-,66-48-,67-53-/t68-,69-,70+,71+,73-,74+,75+,77+,78+,79+,86+/m0/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | (2R)-4-{[(1S)-5-amino-1-{[(1S)-1-{[(1R)-1-carboxyethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}pentyl]-C-hydroxycarbonimidoyl}-2-{[(2S)-1-hydroxy-2-{[(2R)-1-hydroxy-2-{[(2R,3S,4R,5R,6R)-3-hydroxy-6-{[hydroxy({[hydroxy({[(2Z,6Z,10Z,14Z,18Z,22Z,26Z,30Z,34E,38E)-3,7,11,15,19,23,27,31,35,39,43-undecamethyltetratetraconta-2,6,10,14,18,22,26,30,34,38,42-undecaen-1-yl]oxy})phosphoryl]oxy})phosphoryl]oxy}-5-[(1-hydroxyethylidene)amino]-2-(hydroxymethyl)oxan-4-yl]oxy}propylidene]amino}propylidene]amino}butanoic acid |
|---|
|
Traditional IUPAC Name: |
(2R)-4-{[(1S)-5-amino-1-{[(1S)-1-{[(1R)-1-carboxyethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}pentyl]-C-hydroxycarbonimidoyl}-2-{[(2S)-1-hydroxy-2-{[(2R)-1-hydroxy-2-{[(2R,3S,4R,5R,6R)-3-hydroxy-6-{[hydroxy({hydroxy[(2Z,6Z,10Z,14Z,18Z,22Z,26Z,30Z,34E,38E)-3,7,11,15,19,23,27,31,35,39,43-undecamethyltetratetraconta-2,6,10,14,18,22,26,30,34,38,42-undecaen-1-yl]oxyphosphoryl}oxy)phosphoryl]oxy}-5-[(1-hydroxyethylidene)amino]-2-(hydroxymethyl)oxan-4-yl]oxy}propylidene]amino}propylidene]amino}butanoic acid |
|---|
| SMILES: | NCCCC[C@]([H])(N=C(O)CC[C@@]([H])(N=C(O)[C@@](C)([H])N=C(O)[C@](C)([H])O[C@@]1([H])[C@@](O)([H])[C@@]([H])(CO)O[C@]([H])(OP(O)(=O)OP(O)(=O)OC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(\C)CC\C([H])=C(/C)CC\C([H])=C(/C)CCC=C(C)C)[C@]1([H])N=C(C)O)C(=O)O)C(O)=N[C@](C)([H])C(O)=N[C@@](C)([H])C(=O)O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as polyprenyl phospho carbohydrates. These are polyprenyl phosphates with a carbohydrate moiety attached to it. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Prenol lipids |
|---|
| Sub Class | Polyprenols |
|---|
|
Direct Parent |
Polyprenyl phospho carbohydrates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Polyterpenoid
- Bactoprenol diphosphate
- Polyprenyl phospho carbohydrate
- Alpha peptide
- N-acyl-alpha-hexosamine
- N-acyl-aliphatic-alpha amino acid
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Glucosamine
- Amino sugar
- Organic pyrophosphate
- Monosaccharide phosphate
- Isoprenoid phosphate
- Alpha-amino acid or derivatives
- Monoalkyl phosphate
- Amino saccharide
- Alkyl phosphate
- Phosphoric acid ester
- Oxane
- Organic phosphoric acid derivative
- Organic phosphate
- Monosaccharide
- Dicarboxylic acid or derivatives
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Ether
- Dialkyl ether
- Carboxylic acid
- Carboxylic acid derivative
- Carboximidic acid derivative
- Carboximidic acid
- Hydrocarbon derivative
- Primary amine
- Primary alcohol
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Carbonyl group
- Amine
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -3 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Membrane |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | Not Available | | Kegg ID | Not Available | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|