|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001713 |
|---|
|
Identification |
|---|
| Name: |
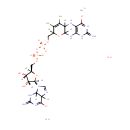
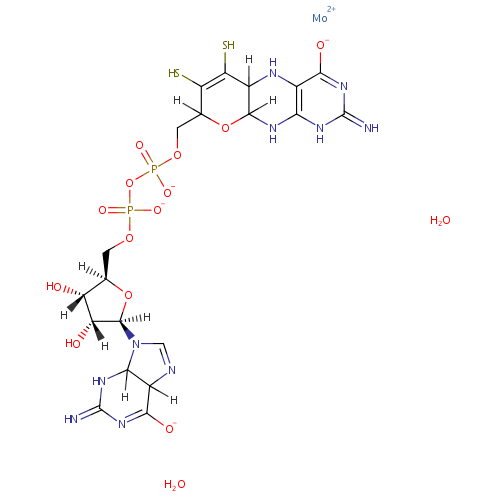
Bis-molybdopterin guanine dinucleotide |
|---|
| Description: | Bis-molybdopterin guanine dinucleotide is a member of the chemical class known as Purine Ribonucleoside Diphosphates. These are purine ribobucleotides with diphosphate group linked to the ribose moiety. |
|---|
|
Structure |
|
|---|
| Synonyms: | - bis-molybdopterin guanine dinucleotide
- MGD
- Molybdopterin guanine dinucleotide
- MoO2(OH)Dtpp-mGDP
- MoO2(OH)Dtpp-mGDP
- MPT-GMP
|
|---|
|
Chemical Formula: |
C20H28MoN10O15P2S2 |
|---|
| Average Molecular Weight: |
870.51 |
|---|
| Monoisotopic Molecular
Weight: |
871.970632526 |
|---|
| InChI Key: |
PNPXRABSZIPVFU-UHFFFAOYSA-J |
|---|
| InChI: | InChI=1S/C20H28N10O13P2S2.Mo.2H2O/c21-19-26-13-7(15(33)28-19)24-6-12(47)11(46)5(41-17(6)25-13)2-40-45(37,38)43-44(35,36)39-1-4-9(31)10(32)18(42-4)30-3-23-8-14(30)27-20(22)29-16(8)34;;;/h3-6,8-10,14,17-18,24,31-32,46-47H,1-2H2,(H,35,36)(H,37,38)(H3,22,27,29,34)(H4,21,25,26,28,33);;2*1H2/q;+2;;/p-4 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | molybdenum(2+) ion 9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[({[({2-imino-4-oxido-6,7-disulfanyl-1H,2H,5H,5aH,8H,9aH,10H-pyrano[3,2-g]pteridin-8-yl}methyl phosphonato)oxy]phosphinato}oxy)methyl]oxolan-2-yl]-2-imino-3,4,5,9-tetrahydro-2H-purin-6-olate dihydrate |
|---|
|
Traditional IUPAC Name: |
molybdenum(2+) ion 9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-({[({2-imino-4-oxido-6,7-disulfanyl-1H,5H,5aH,8H,9aH,10H-pyrano[3,2-g]pteridin-8-yl}methyl phosphonato)oxyphosphinato]oxy}methyl)oxolan-2-yl]-2-imino-4,5-dihydro-3H-purin-6-olate dihydrate |
|---|
| SMILES: | O.O.[Mo++].[H][C@]1(COP([O-])(=O)OP([O-])(=O)OCC2([H])OC3([H])NC4=C(NC3([H])C(S)=C2S)C([O-])=NC(=N)N4)O[C@@]([H])(N2C=NC3([H])C([O-])=NC(=N)NC23[H])[C@]([H])(O)[C@]1([H])O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as purine ribonucleoside diphosphates. These are purine ribobucleotides with diphosphate group linked to the ribose moiety. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
|
Class |
Purine nucleotides |
|---|
| Sub Class | Purine ribonucleotides |
|---|
|
Direct Parent |
Purine ribonucleoside diphosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Purine ribonucleoside diphosphate
- Pyranopterin
- Pterin
- N-glycosyl compound
- Glycosyl compound
- Pteridine
- Organic pyrophosphate
- Monosaccharide phosphate
- Imidazopyrimidine
- Secondary aliphatic/aromatic amine
- Aminopyrimidine
- Alkyl phosphate
- Pyrimidine
- Pyran
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- Monosaccharide
- 1,2,5,6-tetrahydropyrimidine
- Hydropyrimidine
- Saccharide
- Heteroaromatic compound
- Oxolane
- 2-imidazoline
- Tertiary amine
- Secondary alcohol
- Guanidine
- 1,2-diol
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Carboximidamide
- Thioenol
- Secondary amine
- Carboxylic acid amidine
- Amidine
- Alkylthiol
- Hydrocarbon derivative
- Organic transition metal salt
- Organic salt
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Imine
- Amine
- Alcohol
- Organic cation
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aromatic heteropolycyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | 50986239 | | Kegg ID | Not Available | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | CPD-582 | | EcoCyc ID | CPD-582 |
|
|---|