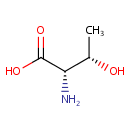
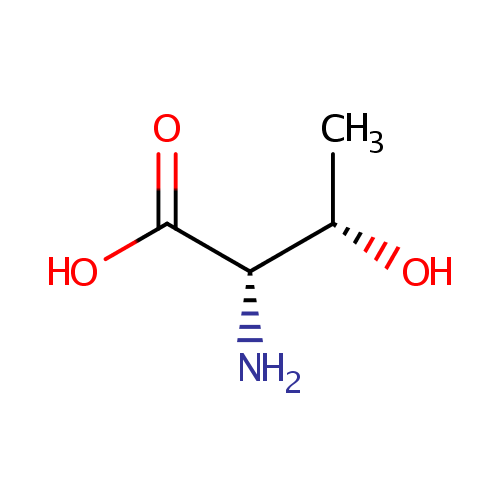
| References: |
- Baier LJ, Sacchettini JC, Knowler WC, Eads J, Paolisso G, Tataranni PA, Mochizuki H, Bennett PH, Bogardus C, Prochazka M: An amino acid substitution in the human intestinal fatty acid binding protein is associated with increased fatty acid binding, increased fat oxidation, and insulin resistance. J Clin Invest. 1995 Mar;95(3):1281-7. Pubmed: 7883976
- Brancati F, Valente EM, Davies NP, Sarkozy A, Sweeney MG, LoMonaco M, Pizzuti A, Hanna MG, Dallapiccola B: Severe infantile hyperkalaemic periodic paralysis and paramyotonia congenita: broadening the clinical spectrum associated with the T704M mutation in SCN4A. J Neurol Neurosurg Psychiatry. 2003 Sep;74(9):1339-41. Pubmed: 12933953
- Contestabile R, Angelaccio S, Bossa F, Wright HT, Scarsdale N, Kazanina G, Schirch V: Role of tyrosine 65 in the mechanism of serine hydroxymethyltransferase. Biochemistry. 2000 Jun 27;39(25):7492-500. Pubmed: 10858298
- Elliot TA, Cree MG, Sanford AP, Wolfe RR, Tipton KD: Milk ingestion stimulates net muscle protein synthesis following resistance exercise. Med Sci Sports Exerc. 2006 Apr;38(4):667-74. Pubmed: 16679981
- Farhana L, Dawson MI, Huang Y, Zhang Y, Rishi AK, Reddy KB, Freeman RS, Fontana JA: Apoptosis signaling by the novel compound 3-Cl-AHPC involves increased EGFR proteolysis and accompanying decreased phosphatidylinositol 3-kinase and AKT kinase activities. Oncogene. 2004 Mar 11;23(10):1874-84. Pubmed: 14981538
- Fiehn O, Kopka J, Trethewey RN, Willmitzer L: Identification of uncommon plant metabolites based on calculation of elemental compositions using gas chromatography and quadrupole mass spectrometry. Anal Chem. 2000 Aug 1;72(15):3573-80. Pubmed: 10952545
- Greeve J, Altkemper I, Dieterich JH, Greten H, Windler E: Apolipoprotein B mRNA editing in 12 different mammalian species: hepatic expression is reflected in low concentrations of apoB-containing plasma lipoproteins. J Lipid Res. 1993 Aug;34(8):1367-83. Pubmed: 8409768
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- Le Floc'h N, Obled C, Seve B: In vivo threonine oxidation rate is dependent on threonine dietary supply in growing pigs fed low to adequate levels. J Nutr. 1995 Oct;125(10):2550-62. Pubmed: 7562090
- Martini C, Trincavelli ML, Tuscano D, Carmassi C, Ciapparelli A, Lucacchini A, Cassano GB, Dell'Osso L: Serotonin-mediated phosphorylation of extracellular regulated kinases in platelets of patients with panic disorder versus controls. Neurochem Int. 2004 Jun;44(8):627-39. Pubmed: 15016478
- Meyer W, Poehling HM, Neurand K: Intraepidermal distribution of free amino acids in porcine skin. J Dermatol Sci. 1991 Sep;2(5):383-92. Pubmed: 1742249
- Nanda N, Bao M, Lin H, Clauser K, Komuves L, Quertermous T, Conley PB, Phillips DR, Hart MJ: Platelet endothelial aggregation receptor 1 (PEAR1), a novel epidermal growth factor repeat-containing transmembrane receptor, participates in platelet contact-induced activation. J Biol Chem. 2005 Jul 1;280(26):24680-9. Epub 2005 Apr 25. Pubmed: 15851471
- Parimi PS, Gruca LL, Kalhan SC: Metabolism of threonine in newborn infants. Am J Physiol Endocrinol Metab. 2005 Dec;289(6):E981-5. Epub 2005 Jul 19. Pubmed: 16030066
- Persson B, Zigler JS Jr, Jornvall H: A super-family of medium-chain dehydrogenases/reductases (MDR). Sub-lines including zeta-crystallin, alcohol and polyol dehydrogenases, quinone oxidoreductase enoyl reductases, VAT-1 and other proteins. Eur J Biochem. 1994 Nov 15;226(1):15-22. Pubmed: 7957243
- Saitoh T, Takemura S, Ueda K, Hosoya H, Nagayama M, Haga H, Kawabata K, Yamagishi A, Takahashi M: Differential localization of non-muscle myosin II isoforms and phosphorylated regulatory light chains in human MRC-5 fibroblasts. FEBS Lett. 2001 Dec 14;509(3):365-9. Pubmed: 11749957
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. Pubmed: 19212411
- van der Werf, M. J., Overkamp, K. M., Muilwijk, B., Coulier, L., Hankemeier, T. (2007). "Microbial metabolomics: toward a platform with full metabolome coverage." Anal Biochem 370:17-25. Pubmed: 17765195
- Winder, C. L., Dunn, W. B., Schuler, S., Broadhurst, D., Jarvis, R., Stephens, G. M., Goodacre, R. (2008). "Global metabolic profiling of Escherichia coli cultures: an evaluation of methods for quenching and extraction of intracellular metabolites." Anal Chem 80:2939-2948. Pubmed: 18331064
- Wishart MJ, Denu JM, Williams JA, Dixon JE: A single mutation converts a novel phosphotyrosine binding domain into a dual-specificity phosphatase. J Biol Chem. 1995 Nov 10;270(45):26782-5. Pubmed: 7592916
|
|---|