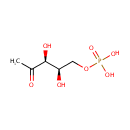
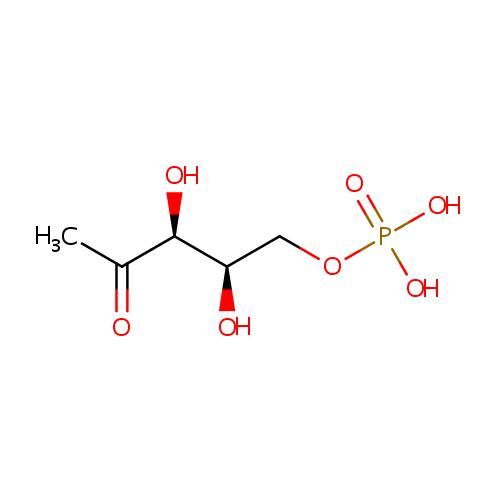
1-Deoxy-D-xylulose 5-phosphate (PAMDB000301)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000301 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | 1-Deoxy-D-xylulose 5-phosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | 1-Deoxy-D-xylulose 5-phosphate is an intermediate in the non-mevalonate pathway. This is an alternative metabolic pathway leading to the formation of isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP). The key enzyme in the non-mevalonate pathway of isoprenoid biosynthesis is 1-deoxy-D-xylulose 5-phosphate reductoisomerase (DXR). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C5H11O7P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 214.1104 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 214.024239218 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | AJPADPZSRRUGHI-RFZPGFLSSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C5H11O7P/c1-3(6)5(8)4(7)2-12-13(9,10)11/h4-5,7-8H,2H2,1H3,(H2,9,10,11)/t4-,5-/m1/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 190079-18-6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | {[(2R,3S)-2,3-dihydroxy-4-oxopentyl]oxy}phosphonic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | 1-deoxy-D-xylulose-5-phosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | CC(=O)[C@@H](O)[C@H](O)COP(O)(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as pentose phosphates. These are carbohydrate derivatives containing a pentose substituted by one or more phosphate groups. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organooxygen compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carbohydrates and carbohydrate conjugates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Monosaccharides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Pentose phosphates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | 1-Deoxy-D-xylulose 5-phosphate + NAD + O-Phospho-4-hydroxy-L-threonine > Carbon dioxide + Hydrogen ion +2 Water + NADH + Pyridoxine 5'-phosphate + Phosphate Adenosine triphosphate + Dehydroglycine + 1-Deoxy-D-xylulose 5-phosphate + Hydrogen ion + IscS with bound sulfur + NADPH > 4-Methyl-5-(2-phosphoethyl)-thiazole + Adenosine monophosphate + Carbon dioxide +2 Water + IscS sulfur acceptor protein + NADP + Pyrophosphate 1-Deoxy-D-xylulose 5-phosphate + Hydrogen ion + NADPH <> 2-C-Methyl-D-erythritol-4-phosphate + NADP D-Glyceraldehyde 3-phosphate + Hydrogen ion + Pyruvic acid <> Carbon dioxide + 1-Deoxy-D-xylulose 5-phosphate Pyruvic acid + D-Glyceraldehyde 3-phosphate <> 1-Deoxy-D-xylulose 5-phosphate + Carbon dioxide 3-Amino-2-oxopropyl phosphate + 1-Deoxy-D-xylulose 5-phosphate + 3-Amino-2-oxopropyl phosphate <> Pyridoxine 5'-phosphate + Phosphate +2 Water 1-Amino-propan-2-one-3-phosphate + 1-Deoxy-D-xylulose 5-phosphate > Hydrogen ion + Pyridoxine 5'-phosphate + Phosphate + Water Adenosine triphosphate + 1-Deoxy-D-xylulose > Hydrogen ion + 1-Deoxy-D-xylulose 5-phosphate + ADP 1-Deoxy-D-xylulose 5-phosphate + 2-iminoacetate + thiocarboxy-adenylate-[sulfur-carrier protein ThiS] > 2-((2R,5Z)-2-Carboxy-4-methylthiazol-5(2H)-ylidene)ethyl phosphate + [sulfur-carrier protein ThiS] +2 Water 1-Deoxy-D-xylulose 5-phosphate + 2-iminoacetate <> 2-[(2R,5Z)-2-Carboxy-4-methylthiazol-5(2H)-ylidene]ethyl phosphate +2 Water 1-Deoxy-D-xylulose 5-phosphate + 2-Amino-3-phosphonopropionic acid + 1-Deoxy-D-xylulose 5-phosphate + 2-Amino-3-phosphonopropionic acid > Pyridoxine 5'-phosphate + Phosphate + Hydrogen ion +2 Water D-Glyceraldehyde 3-phosphate + Pyruvic acid + Hydrogen ion + D-Glyceraldehyde 3-phosphate > 1-Deoxy-D-xylulose 5-phosphate + Carbon dioxide + 1-Deoxy-D-xylulose 5-phosphate D-Glyceraldehyde 3-phosphate + Hydrogen ion + D-Glyceraldehyde 3-phosphate > Carbon dioxide + 1-Deoxy-D-xylulose 5-phosphate + 1-Deoxy-D-xylulose 5-phosphate NADPH + Hydrogen ion + 1-Deoxy-D-xylulose 5-phosphate + NADPH + 1-Deoxy-D-xylulose 5-phosphate > NADP + 2-C-methyl-D-erythritol 4-phosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Blagg, Brian S. J.; Poulter, C. Dale. Synthesis of 1-Deoxy-D-xylulose and 1-Deoxy-D-xylulose-5-phosphate. Journal of Organic Chemistry (1999), 64(5), 1508-1511. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||